HONG KONG, Jun 5, 2022 – (ACN Newswire) – Professor Elaine YK Chow from the Faculty of Medicine at The Chinese University of Hong Kong has received the "2022 Women's Interprofessional Network of the American Diabetes Association Abstract Award" in the category of Clinical Diabetes, Epidemiology, and Diabetes Complications in recognition of her outstanding results and significant contributions in the SENSITIZE study and the whole field of diabetes research. The WIN ADA abstract award category of Clinical Diabetes, Epidemiology, and Diabetes Complications is an important part of WIN ADA awards, voted by WIN ADA Advisory Group, consisting of expert clinicians, scientists, educators, public health practitioners, and other diabetes professionals. It honours women who have made significant contributions to the diabetes research, clinical care, public health, education, and related areas worldwide. The award was presented to Professor Elaine on June 3, 2022.
Professor Elaine will present a clinical study called SENSITIZE, which explored the effects of dorzagliatin, the global first-in-class drug, glucokinase activator (GKA) at the ADA Annual Scientific Sessions. This study has been initiated by Professor Juliana Chan, an international endocrinologist from the Chinese University of Hong Kong, explored the effects of dorzagliatin on patients with recent onset T2D and glucokinase-maturity-onset diabetes of the young (GCK-MODY or MODY-2).
Copyright 2022 ACN Newswire. All rights reserved. http://www.acnnewswire.com
Tag: BioTech
Connected Transactions Involving Subscription by Vivo Suzhou Fund and Centerlab for TOT BIOPHARM Shares
HONG KONG, Jun 2, 2022 – (ACN Newswire) – On June 1, 2022, TOT BIOPHARM (Stock code: 1875. HK) announced that the company entered into a Share Subscription Agreement with VIVO Suzhou Fund and Centerlab. Pursuant to the agreement, Vivo Suzhou Fund and Centerlab agreed to subscribe for 116,250,000 and 33,750,000 shares of TOT BIOPHARM, respectively, at a subscription price of HK$3.15 per share (Premium issuance based on the past 5 trading days) , with a total of 150,000,000 shares, accounting for approximately 24.38% of the company's issued capital as of the date of this announcement. After the completion of the subscription agreement, assuming that all outstanding share options under the company's Pre-IPO Share Option Scheme will not be exercised before completion, the shareholding ratio of Vivo Suzhou Fund and Centerlab will reach approximately 28.68% and 28.66%.
In recent years, TOT BIOPHARM has accelerated its strategic adjustment, strengthened its competitive advantage in the antibody conjugated drugs (ADC) field, and facilitated the construction of an industry-leading ADC one-stop commercialization platform. The company has made every effort to expand its one-stop innovative drug CDMO business, achieving breakthrough results and an ever-growing business scale. This business has already become a new engine for corporate growth.
The net proceeds from the Subscriptions have amounted to HK$470 million (approximately RMB 400 million), which will improve corporate liquidity, expand the capital base, optimize the capital structure, and provide support for corporate long-term growth. The company intends to apply the proceeds to: further expand CDMO business and strengthen project cooperation with domestic and foreign pharmaceutical companies; promote the continuous construction of global R&D centers, improving ADC commercialization capacities, thereby improving product cost-effectiveness; complete the phase III clinical trial of TAA013and etc.; carry out production and marketing of Pusintin, Tazian, and Megaxia, etc.
Mr. Fu Shan, Chairman of TOT BIOPHARM, Managing Partner, Global Co-CEO and Asia CEO of Vivo Capital LLC, highlighted: "This transaction is another major milestone of TOT BIOPHARM development, which symbolizes its clear strategic planning and business performance have been highly recognized by shareholders. Over the past years, the management team of the company has always concentrated on the advantages of core competencies, optimized resource allocation, and built a differentiated pathway to create a domestically leading and internationally competitive ADC platform. They have also actively expanded CDMO business and established a commercialization platform with great competitive edges and industrial value, showing strong potential and sustainability, which gains shareholders and the board of directors' confidence in the company's future development!"
Dr. Liu Jun, CEO of TOT BIOPHARM, expressed: "It is a great honor to gain the strong support and recognition of our two major shareholders, Vivo Capital and Centerlab, during the critical period of us re-setting corporate priorities on commercialization and strategic adjustment with ADC as the core! By far, our strategic transformation and up-gradation to ADC CDMO have yielded remarkable results. This capital increase will further enhance our capital strength and provide financial support for corporate sustainability. In the upcoming days, we will continue take efforts to optimize capital structure, establish a diversified and sustainable business model, as well as to enhance the comprehensive strength of process development, accelerate the ADC commercialization process, and create a new engine for CDMO business growth, thereby strengthening the corporate earning capacity, and creating bountiful returns for shareholders and investors."
About Vivo Suzhou Fund and Other Funds Managed by Vivo Capital LLC
Vivo Suzhou Fund is a limited partnership established in the PRC managed by Vivo Equity Investment Management (Shanghai) Co., Ltd. , a private equity fund manager registered with the Asset Management Association of China which is directly wholly-owned and controlled by Vivo Capital LLC. Vivo VIII Funds, which are also funds managed by Vivo Capital LLC, have been shareholders of the Company since December 2015. Vivo Capital LLC's predecessor was founded in the United States in 1996. Vivo Capital LLC was incorporated in the State of California of the United States in 2005, and is an investment adviser registered with the United States Securities and Exchange Commission. Spanning private equity, public equity and venture capital, it is a global healthcare investment firm that focuses on finding and building value in healthcare companies in the United States and Greater China. It invests in both public and private healthcare companies through funds managed by it or its affiliate, including Vivo Suzhou Fund and Vivo VIII Funds, among others. In terms of assets under management (AUM), as of 31 December 2021, Vivo Capital LLC managed over US$6.4 billion in client assets on a discretionary basis.
About Centerlab
Centerlab was incorporated in Taiwan in 1959 and its shares were listed on the Taipei Exchange in 2003 (stock code: 4123). It is Taiwan's largest manufacturer of oral liquid pharmaceuticals, with a market share of about 70%. Its products are mainly oral solutions, including syrups, suspensions, emulsions and other dosage forms. It also engages in new drug research and development, active pharmaceutical ingredients, minimally invasive medical devices and other businesses through investments in other corporations. In recent years, Centerlab is known as the "Biotech Industrial Bank" in Taiwan for its track record of incubating or investing in various biotech and related companies listed or to be listed on the Stock Exchange or the Taipei Exchange (including the Company) and fostering their growth and development. As of 31 December 2021, Centerlab had total assets of NT$30.4 billion (equivalent to approximately US$1.1 billion). Centerlab (together with its former associate BioEngine Venture Capital Inc. and/or current associate BioEngine Technology) has been a shareholder of the Company since January 2011.
Forward-Looking Statements
This news release may contain certain forward-looking statements that are, by their nature, subject to significant risks and uncertainties. The words "anticipate", "believe", "estimate", "expect", "intend" and similar expressions, as they relate to TOT BIOPHARM, are intended to identify certain of such forward-looking statements. TOT BIOPHARM does not intend to update these forward-looking statements regularly.
These forward-looking statements are based on the existing beliefs, assumptions, expectations, estimates, projections and understandings of the management of TOT BIOPHARM with respect to future events at the time these statements are made. These statements are not a guarantee of future developments and are subject to risks, uncertainties and other factors, some of which are beyond TOT BIOPHARM's control and are difficult to predict. Consequently, actual results may differ materially from information contained in the forward-looking statements as a result of future changes or developments in our business, TOT BIOPHARM's competitive environment and political, economic, legal and social conditions.
TOT BIOPHARM, the Directors and the employees of TOT BIOPHARM assume (a) no obligation to correct or update the forward-looking statements contained in this site; and (b) no liability in the event that any of the forward-looking statements does not materialize or turn out to be incorrect.
Copyright 2022 ACN Newswire. All rights reserved. http://www.acnnewswire.com
Boehringer Ingelheim Enters Global Licensing Agreement to Develop and Commercialize Innovative Antibodies from A*STAR for Targeted Cancer Therapies

Ingelheim, Germany and Singapore, Jun 2, 2022 – (ACN Newswire) – Boehringer Ingelheim and the Agency for Science, Technology and Research (A*STAR) today announced a global licensing agreement under which Boehringer Ingelheim will obtain exclusive worldwide rights to research, develop and commercialize products based on a panel of innovative, tumor-specific antibodies from A*STAR. Boehringer Ingelheim aims to use these antibodies to direct therapeutic effector mechanisms such as antibody-drug conjugates (ADCs) and T-cell engagers exclusively to tumor cells, and to that end develop a range of highly targeted cancer treatments.
 |
"Boehringer Ingelheim believes that these promising antibodies in-licensed from A*STAR will help us advance potent therapeutic candidates against key molecular cancer targets," says Clive R. Wood, Senior Corporate Vice President and Global Head of Discovery Research at Boehringer Ingelheim and continues: "We look forward to developing these assets for a broad range of cancers with the goal to deliver breakthrough opportunities for patients."
Boehringer Ingelheim is pioneering a range of versatile therapeutic platforms in order to develop innovative medicines that target the tumor directly (tumor cell-directed therapies) or that enable the immune system to target the tumor (immune cell-targeted therapies). One tumor cell-directed modality is antibody drug conjugates, which allows for delivery of toxins directly into tumor cells. Another is T-cell engagers facilitating direct contact between T-cells and tumor cells, leading to T-cell-mediated killing of the tumor. Both technologies are directed towards markers on the surface of cancer cells, also known as antigens, in order to attack tumor cells but spare healthy tissues.
The innovative antibodies from A*STAR can potentially enable the development of safer, more efficacious therapies as they selectively bind to antigens that are highly expressed on tumor cells but are absent on normal healthy tissues.
Under the terms of the agreement, Boehringer Ingelheim will be responsible for further research, preclinical and clinical development as well as commercialization of targeted cancer therapies using the antibodies from A*STAR. A*STAR may receive payments totalling >100 million EUR in upfront and success-based development and commercialization milestones.
The technology used to identify the unique A*STAR antibodies resulted from a multi-institutional collaboration in Singapore. A*STAR's Genome Institute of Singapore (GIS) and Institute of Bioengineering & Bioimaging (IBB) generated antibodies which exclusively target antigens that were initially identified from gastric cancer cells. Experimental Drug Development Centre (EDDC), Singapore's national platform for drug discovery and development hosted by A*STAR, then optimized the antibodies and confirmed their applicability to a range of other solid cancers. EDDC also demonstrated the utility of these antibodies in directing different therapeutic modalities selectively to cancer cells.
Professor Damian O'Connell, Chief Executive Officer of EDDC, says, "As Singapore's national drug discovery and development platform, EDDC is proud to translate great science in Singapore into valuable assets that can enable the precision treatment of cancer. We believe that Boehringer Ingelheim, with its broad expertise and technologies, is the right partner to maximize the potential of these antibodies for the development of safer, more targeted therapies for cancer patients."
Professor Tan Sze Wee, Assistant Chief Executive (Enterprise) of A*STAR, says, "These antibodies were developed in Singapore through close collaboration across multiple institutions. There was also strong support by the Singapore Gastric Cancer Consortium. The agreement is testament to the best-in-class research taking place in Singapore. Cancer is a devastating disease, and we hope the fruits of our research can improve patient outcomes."
ANNEX A – Innovative antibody enables targeted treatment of solid tumours (Infographic)
ANNEX B – About the Research Partners in Singapore
See: https://www.acnnewswire.com/docs/Multimedia/ASTAR-BI.pdf
For media queries and clarifications, please contact:
Dr. Reinhard Malin
Innovation Unit Communications
Corporate Affairs
Boehringer Ingelheim
Tel: +49 6132 77-90815
Email: reinhard.malin@boehringeringelheim.com
Owen Sia
Assistant Head, Corporate Communications
Agency for Science, Technology and Research
Tel: +65 6517 7866
Email: owen_sia@hq.a-star.edu.sg
About Boehringer Ingelheim
Boehringer Ingelheim is working on breakthrough therapies that improve the lives of humans and animals. As a leading research-driven biopharmaceutical company, the company creates value through innovation in areas of high unmet medical need. Around 52,000 employees serve more than 130 markets in the three business areas, Human Pharma, Animal Health, and Biopharmaceutical Contract Manufacturing. Learn more at www.boehringer-ingelheim.com.
Boehringer Ingelheim in Oncology
Boehringer Ingelheim is a family-owned company committed to pioneering treatment advancements in some of the most challenging and most impactful areas of cancer. Our goal is to transform the lives of people with cancer by delivering life-enhancing innovations in areas of high unmet need. Our aspiration is to deliver faster and better treatment options for people with cancer today, tomorrow and beyond. Our rich pipeline of next-generation oncology treatments is comprised of cancer cell-directed and immuno-oncology therapies, the smart combinations of which may hold the greatest benefit for most patients.
In immuno-oncology, we aim to turn poorly immunogenic "cold" tumors into immunogenic "hot" tumors through complementary platforms, including Antibody Drug Conjugates (ADCs), T Cell Engagers (TCEs), oncolytic viruses, and cancer vaccines. For cancer cell-targeted treatments, we address the key drivers of cancer, e.g., KRAS, p53, Beta-catenin and MYC, where there has been no hope for patients for a long time.
We proactively collaborate with patients, advocacy organizations, the world's leading academic and industry, because we believe together, we can make a bigger impact in transforming the lives of people with cancer.
About the Agency for Science, Technology and Research (A*STAR)
The Agency for Science, Technology and Research (A*STAR) is Singapore's lead public sector R&D agency. Through open innovation, we collaborate with our partners in both the public and private sectors to benefit the economy and society. As a Science and Technology Organisation, A*STAR bridges the gap between academia and industry. Our research creates economic growth and jobs for Singapore, and enhances lives by improving societal outcomes in healthcare, urban living, and sustainability. A*STAR plays a key role in nurturing scientific talent and leaders for the wider research community and industry. A*STAR's R&D activities span biomedical sciences to physical sciences and engineering, with research entities primarily located in Biopolis and Fusionopolis. For ongoing news, visit www.a-star.edu.sg.
Copyright 2022 ACN Newswire. All rights reserved. http://www.acnnewswire.com
Novotech Sponsors Pre-ASCO China Summit Expert Panels on Early Phase Oncology Trials and Regulatory Strategy for China and US

SYDNEY, Jun 2, 2022 – (ACN Newswire) – Novotech, the leading Asia Pacific biotech specialist CRO which has recently expanded its services to the US, has sponsored expert panels at the Pre-ASCO China Summit 2022 bringing together leaders in oncology drug development in China and the US. The Pre-ASCO series was watched by more than 20,000 people globally.
The Novotech sponsored Summit sessions are:
– Go/No Go Decisions Based on Early Phase Oncology Trials – Watch Here https://tinyurl.com/novotech-webinars
– Development and Regulatory Strategy for China & US – Watch Here https://tinyurl.com/novotech-whitepapers
China offers a compelling solution for expedited clinical trials especially in oncology with its vast patient populations and world-class KOLs.
– According to Global Data whitepaper: EVOLUTION OF CLINICAL TRIALS IN THE ASIA PACIFIC REGION COMPARED TO THE US AND THE EU5 https://tinyurl.com/novotech-whitepapers
– Over the past 5 years, growth of oncology trials (CAGR of 25%) in China outpaced other countries.
– This growth was associated with a significant increase in cancer incidence, development of innovative products by domestic companies, focus on immuno-oncology, and leadership in cell therapy
– China also recorded the largest number of new trials, followed by the US. In China, the number of new trials doubled during 2017-2021. Notably, China's regulatory reforms have accelerated drug approval by reducing the overall review and process.
– In addition, over the last five years more 70,000 new clinical trials were registered in the APAC region, the US, and the EU5. The APAC region was the largest contributor, with more than 50% of the trials followed by the US (29%) and the EU5 (17%).
– The APAC region has become the preferred destination for conducting clinical trials due to its large patient population, ease of regulatory compliance, low cost of conducting studies, high-quality standards and the presence of top clinical sites.
Earlier this month, Novotech announced the acquisition of US CRO, NCGS as part of a US service expansion program for its global base of clients.
Novotech, which has a reputation for delivering full-service, high-quality expedited clinical trials in Asia-Pacific, can now offer its biotech clients clinical services in the US to support later phase global studies.
NCGS was established in 1984 in South Carolina and has a workforce of about 300 professionals across the US.
Novotech CEO Dr. John Moller said:
"This is a strategic move to provide US-based expertise and infrastructure for our US clients wanting trials in APAC and the US, and for our APAC clients wanting US clinical programs."
"Clients will receive seamless service, with a unified approach to systems and SOPs," Moller said.
Novotech has recently been benchmarked as a top 10 CRO among the world's leading CROs, and has signed 45 Leading Site Partnership agreements over the last 3 years.
With more than 2,000 highly experienced professionals, clients benefit from strong site and Key Opinion Leader relationships, deep regulatory expertise, and the ability to accelerate clinical trials across the Asia Pacific and in particular China.
Novotech recently announced a partnership and investment in tech firm Prospection to support accelerated clinical trials with healthcare data analytics using real-world data.
Novotech has also partnered with virtual research organisation ObvioHealth to expedite trials leveraging remote technologies. This patient-centric approach improves subject retention and allows sponsors to reach populations outside major cities for clinical trial participation.
About Novotech Health Holdings
Novotech Health Holdings Pte. Ltd. ("Novotech") is a leading Asia-Pacific biotech specialist CRO and consists of two operating brands, Novotech and PPC. Novotech is a CRO with integrated labs and phase I facilities providing drug development consulting and clinical development services. It has been instrumental in the success of approximately 3,700 clinical trials across all trial phases and broad range of therapeutic areas. Novotech is well positioned to serve biopharmaceutical clients conducting clinical trials in Asia and globally. For more information visit https://novotech-cro.com/contact
Media Contact
David James
communications@novotech-cro.com
AU: +61 2 8218 2144
USA: +1 415 951 3228
Asia: +65 3159 3427
Copyright 2022 ACN Newswire. All rights reserved. http://www.acnnewswire.com
Connected Transactions Involving Subscription by Vivo Suzhou Fund and Centerlab for TOP BIOPHARM Shares
HONG KONG, Jun 1, 2022 – (ACN Newswire) – TOT BIOPHARM (Stock code: 1875. HK) announced that the company entered into a Share Subscription Agreement with VIVO Suzhou Fund and Centerlab. Pursuant to the agreement, Vivo Suzhou Fund and Centerlab agreed to subscribe for 116,250,000 and 33,750,000 shares of TOT BIOPHARM, respectively, at a subscription price of HK$3.15 per share (Premium issuance based on the past 5 trading days) , with a total of 150,000,000 shares, accounting for approximately 24.38% of the company's issued capital as of the date of this announcement. After the completion of the subscription agreement, assuming that all outstanding share options under the company's Pre-IPO Share Option Scheme will not be exercised before completion, the shareholding ratio of Vivo Suzhou Fund and Centerlab will reach approximately 28.68% and 28.66%.
In recent years, TOT BIOPHARM has accelerated its strategic adjustment, strengthened its competitive advantage in the antibody conjugated drugs (ADC) field, and facilitated the construction of an industry-leading ADC one-stop commercialization platform. The company has made every effort to expand its one-stop innovative drug CDMO business, achieving breakthrough results and an ever-growing business scale. This business has already become a new engine for corporate growth.
The net proceeds from the Subscriptions have amounted to HK$470 million (approximately RMB 400 million), which will improve corporate liquidity, expand the capital base, optimize the capital structure, and provide support for corporate long-term growth. The company intends to apply the proceeds to: further expand CDMO business and strengthen project cooperation with domestic and foreign pharmaceutical companies; promote the continuous construction of global R&D centers, improving ADC commercialization capacities, thereby improving product cost-effectiveness; complete the phase III clinical trial of TAA013and etc.; carry out production and marketing of Pusintin, Tazian, and Megaxia, etc.
Mr. Fu Shan, Chairman of TOT BIOPHARM, Managing Partner, Global Co-CEO and Asia CEO of Vivo Capital LLC, highlighted: "This transaction is another major milestone of TOT BIOPHARM development, which symbolizes its clear strategic planning and business performance have been highly recognized by shareholders. Over the past years, the management team of the company has always concentrated on the advantages of core competencies, optimized resource allocation, and built a differentiated pathway to create a domestically leading and internationally competitive ADC platform. They have also actively expanded CDMO business and established a commercialization platform with great competitive edges and industrial value, showing strong potential and sustainability, which gains shareholders and the board of directors' confidence in the company's future development!"
Dr. Liu Jun, CEO of TOT BIOPHARM, expressed: "It is a great honor to gain the strong support and recognition of our two major shareholders, Vivo Capital and Centerlab, during the critical period of us re-setting corporate priorities on commercialization and strategic adjustment with ADC as the core! By far, our strategic transformation and up-gradation to ADC CDMO have yielded remarkable results. This capital increase will further enhance our capital strength and provide financial support for corporate sustainability. In the upcoming days, we will continue take efforts to optimize capital structure, establish a diversified and sustainable business model, as well as to enhance the comprehensive strength of process development, accelerate the ADC commercialization process, and create a new engine for CDMO business growth, thereby strengthening the corporate earning capacity, and creating bountiful returns for shareholders and investors."
About Vivo Suzhou Fund and Other Funds Managed by Vivo Capital LLC
Vivo Suzhou Fund is a limited partnership established in the PRC managed by Vivo Equity Investment Management (Shanghai) Co., Ltd. , a private equity fund manager registered with the Asset Management Association of China which is directly wholly-owned and controlled by Vivo Capital LLC. Vivo VIII Funds, which are also funds managed by Vivo Capital LLC, have been shareholders of the Company since December 2015. Vivo Capital LLC's predecessor was founded in the United States in 1996. Vivo Capital LLC was incorporated in the State of California of the United States in 2005, and is an investment adviser registered with the United States Securities and Exchange Commission. Spanning private equity, public equity and venture capital, it is a global healthcare investment firm that focuses on finding and building value in healthcare companies in the United States and Greater China. It invests in both public and private healthcare companies through funds managed by it or its affiliate, including Vivo Suzhou Fund and Vivo VIII Funds, among others. In terms of assets under management (AUM), as of 31 December 2021, Vivo Capital LLC managed over US$6.4 billion in client assets on a discretionary basis.
About Centerlab
Centerlab was incorporated in Taiwan in 1959 and its shares were listed on the Taipei Exchange in 2003 (stock code: 4123). It is Taiwan's largest manufacturer of oral liquid pharmaceuticals, with a market share of about 70%. Its products are mainly oral solutions, including syrups, suspensions, emulsions and other dosage forms. It also engages in new drug research and development, active pharmaceutical ingredients, minimally invasive medical devices and other businesses through investments in other corporations. In recent years, Centerlab is known as the "Biotech Industrial Bank" in Taiwan for its track record of incubating or investing in various biotech and related companies listed or to be listed on the Stock Exchange or the Taipei Exchange (including the Company) and fostering their growth and development. As of 31 December 2021, Centerlab had total assets of NT$30.4 billion (equivalent to approximately US$1.1 billion). Centerlab (together with its former associate BioEngine Venture Capital Inc. and/or current associate BioEngine Technology) has been a shareholder of the Company since January 2011.
Forward-Looking Statements
This news release may contain certain forward-looking statements that are, by their nature, subject to significant risks and uncertainties. The words "anticipate", "believe", "estimate", "expect", "intend" and similar expressions, as they relate to TOT BIOPHARM, are intended to identify certain of such forward-looking statements. TOT BIOPHARM does not intend to update these forward-looking statements regularly.
These forward-looking statements are based on the existing beliefs, assumptions, expectations, estimates, projections and understandings of the management of TOT BIOPHARM with respect to future events at the time these statements are made. These statements are not a guarantee of future developments and are subject to risks, uncertainties and other factors, some of which are beyond TOT BIOPHARM's control and are difficult to predict. Consequently, actual results may differ materially from information contained in the forward-looking statements as a result of future changes or developments in our business, TOT BIOPHARM's competitive environment and political, economic, legal and social conditions.
TOT BIOPHARM, the Directors and the employees of TOT BIOPHARM assume (a) no obligation to correct or update the forward-looking statements contained in this site; and (b) no liability in the event that any of the forward-looking statements does not materialize or turn out to be incorrect.
Copyright 2022 ACN Newswire. All rights reserved. http://www.acnnewswire.com
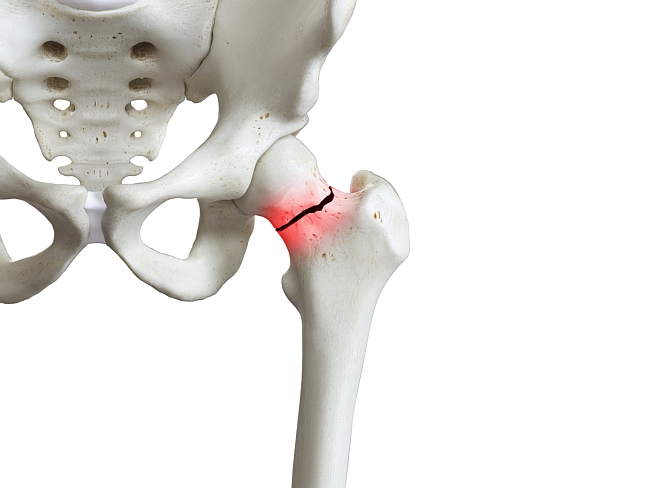
The APCO Bone Health QI Tool Kit – Putting a brake on fractures in the world’s most populous & fastest ageing region
NEW DELHI, May 24 – (ACN Newswire) – An osteoporotic fracture occurs every three seconds worldwide. With more than 50 per cent of the world’s hip fractures expected to occur in the Asia Pacific (AP) region over the next two decades, the Asia Pacific Consortium on Osteoporosis (APCO) has launched a Bone Health QI Tool Kit today to promote quality improvement (QI) of osteoporosis care.
This comprehensive tool kit, that encompasses multiple components in the screening, diagnosis and management of osteoporosis, is a world-first of its kind, said Dr Manju Chandran, APCO Chairperson and Director of the Osteoporosis and Bone Metabolism Unit, Singapore General Hospital, Singapore.
“Previous audits have focused on just a single aspect of osteoporosis care, namely the identification and appropriate management of osteoporotic fragility fractures. A perfect storm of a rise in osteoporosis among Asia-Pacific populations is expected to occur due to the region’s rapidly ageing population, mounting urbanisation, and subsequent increase in sedentary lifestyles,”2 Dr Chandran said.
“A recent analysis of nine Asian countries and regions reveals the number of hip fractures will more than double from 1.13 million in 2018, to 2.54 million in 2050,3 primarily due to changing population demographics.”
The APCO Bone Health QI Tool Kit is based on seven selected standards from The APCO Framework that apply to the clinical setting. Implementation of the QI will be through iterative Plan-Do-Study-Act (PDSA) cycles.
“Once the audits are completed, healthcare professionals (HCPs) and medical centres can commence implementing changes to their clinical practice. APCO is in the process of engaging QI experts to help train APCO members to perform such projects in their individual medical centres and practices,” said Dr Chandran.
The tool kit will enable these auditing exercises to be undertaken to establish levels of adherence with those standards of care within The APCO Framework that can be implemented directly into clinical practice.4
Between 2025 and 2050, the population of India is projected to grow from 1.4 billion, to 1.88 billion, with those aged over 50 years expected to constitute 33 per cent of the population by 2050.5 The number of hip fractures in India is anticipated to more than double, from approximately 332,000 in 2018, to 792,000 in 2050,5 noting hip fractures occur a decade earlier in Indian versus Western populations.3 The projected direct cost of hip fractures is expected to increase from USD 256 million (INR 19,839,618,560), to USD 612 million (INR 47,471,309,388) between 2018 and 2050.3
“The likelihood of successfully creating change to stem this bone health crisis will rest largely upon the efforts of HCPs to engage their peers to work towards shared goals. Articulating the benefits of the APCO Bone Health QI activity can help promote engagement and cooperation,” said Professor Ambrish Mithal, APCO Committee member, Endocrinologist, and Chairperson and Head of Endocrinology and Diabetes, Max Healthcare – Pan-Max, Saket, New Delhi, India.
“The critical factors to success include having a supportive organisational culture, good design and planning, and follow through on results, ensuring the data collection is meaningful and achievable. Data collected through the audit should be used to benchmark practice against the standards recommended by The APCO Framework. This will show stakeholders that the audit was worth the effort, and that it contributed to tangible change,” Prof Mithal said.
The APCO audit focuses on investigating the patient pathway, identifying the gaps or missed opportunities for patients at risk, and then the PDSA activity is used to improve quality of care and to formulate recommendations.
“It is hoped that lessons from implementing these clinical standards at the institutional level will be key to achieving change at the national level, through the development, or revision of national guidelines, and policy change,” said Dr Philippe Halbout, CEO, International Osteoporosis Foundation, Switzerland.
“These coordinated systems of care aim to identify, treat and monitor patients with osteoporosis. Timely intervention can reduce fractures by up to 50 per cent, deliver substantial financial savings, and ultimately, save lives.”
Osteoporosis is greatly under-diagnosed and under-treated in Asia, even among those at highest risk who have already fractured.6
The APCO Bone Health QI Tool Kit focuses on closing this gap by:
* Understanding the baseline variables and current state of osteoporosis care in any given institution or health care practice.
* Enabling benchmarking clinical practice against The APCO Framework, evidence-based clinical practice guidelines or established protocols for the chosen standard of care.
* Using the findings to then improve the quality of care through iterative PDSA cycles.
* Using the lessons learned from implementation of the APCO Clinical Standard at an institutional level, to build momentum and work towards inclusion of the quality improvement/clinical standard in national guidelines, and lobbying for funding and policy change, as appropriate.
Practically, PDSA cycles are a quality improvement strategy that tests a change on a small scale. The PDSA builds on the learning from test cycles in a structured way before implementation on a wider scale.7
The PDSA cycles are iterative. Each cycle involves the following four stages:
1. Plan — developing a plan to test the change;
2. Do — carrying out the test;
3. Study — observing and learning; and
4. Act — planning the next change cycle or full implementation.
“Assessment of adherence to pharmacological treatments that are recommended in new or revised osteoporosis clinical guidelines should be undertaken on an ongoing basis, after initiation of therapy, and appropriate corrective action can be taken if treated individuals have become non-adherent,” Dr Chandran advised.
Every patient living with osteoporosis and/or a fragility fracture passes through several points on their journey from diagnosis, through to treatment and follow-up. The interface of this pathway with other entities, for example, referrals to other care givers or departments, post- discharge follow-ups, are also important considerations in the quality of care received.
“The launch of the APCO Bone Health QI Tool Kit demonstrates the organisation’s ongoing commitment to achieving better care for patients with greater consistency in national and regional clinical practice guidelines for the screening, diagnosis, and management of osteoporosis in the Asia Pacific,” said Dr Halbout.
To learn more about the APCO Bone Health QI Tool Kit, visit www.apcobonehealth.org or follow APCO on LinkedIn: asia-pacific-consortium-on-osteoporosis.
About the APCO Bone Health QI Tool Kit
APCO developed the Bone Health QI Tool Kit to enable healthcare providers worldwide, to benchmark their practices against the clinical standards proposed in The APCO Framework. The tool kit provides guidance on undertaking and implementing quality improvement (QI) projects centred around seven key clinical standards for the screening, diagnosis, and management of osteoporosis.
With the key objective of promoting QI of osteoporosis care, the APCO audit focuses on investigating the patient pathway, identifying the gaps, or missed opportunities for patients at risk, and formulating recommendations to address these gaps. Implementation of the QI will be through Plan-Do-Study-Act (PDSA) cycles – a quality improvement strategy that tests a change on a small scale, before implementation on a wider scale. The tool kit offers practical tips on conducting an audit, that generates meaningful data, that can lead ultimately to implementing changes in clinical practices.
About APCO
The Asia Pacific Consortium on Osteoporosis (APCO) comprises 46 osteoporosis experts from 20 countries and regions, charged with developing tangible solutions to the substantive challenges involving osteoporosis management and fracture prevention in this most populated and fastest growing part of the world. APCO’s mission is to engage with relevant stakeholders, including healthcare providers, policy makers and the public, to help develop and implement country and region-specific programs for the prevention and treatment of osteoporosis, and its complication of fragility fractures, in the Asia Pacific. http://www.apcobonehealth.org
About osteoporosis in the Asia Pacific region
* Osteoporosis – the most common disease – is characterised by porous and brittle bones whose density and quality are poor, resulting in structural skeletal deterioration.1
* The Asia Pacific is home to 4.5 billion people with vastly different healthcare systems.8
* In most developing countries of the Asia Pacific, especially in rural areas, DXA technology is not widely available.9
* Low levels of vitamin D are observed region-wide in the Asia Pacific. This can be attributed to several causal factors, including genetic, lifestyle, seasonal and geographical influences.10
* More than 68 per cent of the countries in the IOF’s Asia Pacific’s Regional Audit now have populations that are over 50 per cent urban, and the resulting reduction in sunlight exposure due to pollution and overcrowding, is affecting vitamin D levels, which contribute to osteoporosis risk.10
* Nearly all Asian countries fall far below The Food and Agriculture Organization of the United Nations / World Health Organization (WHO) recommendations for calcium intake of between 1,000 and 1,300 mg/day. The calcium intake for Malaysia, the Philippines and India remains below 500 mg/day.10
DIGITAL MEDIA KIT : http://www.apcotoolkit.org
Asia Pacific Consortium on Osteoporosis (APCO)
secretariat@apcobonehealth.org
References
1. International Osteoporosis Foundation (IOF). Capture the fracture. [Oct, 2021]; https://www.capturethefracture.org/about.
2. Chandran M, et al., Development of the Asia Pacific Consortium on Osteoporosis (APCO) Framework: clinical standards of care for the screening, diagnosis, and management of osteoporosis in the Asia-Pacific region. Osteoporos Int., 2021. 32(7): p. 1249-1275.
3. Cheung, C.-L., et al., An updated hip fracture projection in Asia: The Asian Federation of Osteoporosis Societies study. Osteoporosis and sarcopenia, 2018. 4(1): p. 16-21.
4. Chandran, M., Ebeling, P., Mitchell, P., Nguyen, T., Harmonization of Osteoporosis Guidelines: Paving the Way for Disrupting the Status Quo in Osteoporosis Management in the Asia Pacific. JBMR, 2022.
5. Watts, J., Ambimanyi-Ochom, J, & Sander K, Osteoporosis costing all Australians: A new burden of disease analysis 2012-2022. 2013, Osteoporosis Australia.
6. International Osteoporosis Foundation (IOF). Key statistics for Asia. [cited October 2021]; https://www.osteoporosis.foundation/facts-statistics/key-statistic-for-asia.
7. NHS England and NHS Improvement. Plan, Do, Study, Act (PDSA) cycles and the model for improvement. https://www.england.nhs.uk/wp-content/uploads/2022/01/qsir-pdsa-cycles-model-for-improvement.pdf.
8. Wang, P., et al., Estimation of Prevalence of Osteoporosis Using OSTA and Its Correlation with Sociodemographic Factors, Disability and Comorbidities. International journal of environmental research and public health, 2019. 16(13): p. 2338.
9. Chandran, M., et al., IQ driving QI: the Asia Pacific Consortium on Osteoporosis (APCO): an innovative and collaborative initiative to improve osteoporosis care in the Asia Pacific. Osteoporos Int, 2020. 31(11): p. 2077-2081.
10. International Osteoporosis Foundation (IOF). The Asia-Pacific Regional Audit. 2013 [cited May 2022]; https://www.osteoporosis.foundation/sites/iofbonehealth/files/2019-06/2013_Asia_Pacific_Audit_English.pdf.
Copyright 2022 ACN Newswire. All rights reserved. http://www.acnnewswire.com
The APCO Bone Health QI Tool Kit – Putting a brake on fractures in the world’s most populous & fastest ageing region
SELANGOR, MALAYSIA, May 24 – (ACN Newswire) – An osteoporotic fracture occurs every three seconds worldwide.1 With more than 50 per cent of the world’s hip fractures expected to occur in the Asia Pacific (AP) region over the next two decades, the Asia Pacific Consortium on Osteoporosis (APCO) has launched a Bone Health QI Tool Kit today (May 24, 2022), to promote quality improvement (QI) of osteoporosis care.
This comprehensive tool kit, that encompasses multiple components in the screening, diagnosis and management of osteoporosis, is a world-first of its kind, said Dr Manju Chandran, APCO Chairperson and Director of the Osteoporosis and Bone Metabolism Unit, Singapore General Hospital, Singapore.
“Previous audits have focused on just a single aspect of osteoporosis care, namely the identification and appropriate management of osteoporotic fragility fractures. A perfect storm of a rise in osteoporosis among Asia-Pacific populations is expected to occur due to the region’s rapidly ageing population, mounting urbanisation, and subsequent increase in sedentary lifestyles,”2 Dr Chandran said.
“A recent analysis of nine Asian countries and regions reveals the number of hip fractures will more than double from 1.13 million in 2018, to 2.54 million in 2050,3 primarily due to changing population demographics.”
The APCO Bone Health QI Tool Kit is based on seven selected standards from The APCO Framework that apply to the clinical setting. Implementation of the QI will be through iterative Plan-Do-Study-Act (PDSA) cycles.
“Once the audits are completed, healthcare professionals (HCPs) and medical centres can commence implementing changes to their clinical practice. APCO is in the process of engaging QI experts to help train APCO members to perform such projects in their individual medical centres and practices,” said Dr Chandran.
The tool kit will enable these auditing exercises to be undertaken to establish levels of adherence with those standards of care within The APCO Framework that can be implemented directly into clinical practice.4
Advancing age, reduced body weight, and minimal physical activity significantly predicts osteoporosis in men, while older age, low body weight, and low-income status significantly predicts osteoporosis in women 5, according to Dr Fen Lee Hew, APCO Committee member, Consultant Physician and Endocrinologist, Puchong Medical Specialist Centre and Subang Jaya Medical Centre (SJMC), Malaysia.
“Concerningly, the Malaysian population is ageing rapidly. Those aged 50 years and over are expected to increase from 5.3 million in 2013, to 13.9 million in 2050.5 Furthermore, Malaysia is expected to experience a 3.55-fold increase in hip fracture incidence by 2050, compared to 2018.5
“The likelihood of successfully creating change to stem this bone health crisis will rest largely upon the efforts of HCPs to engage their peers to work towards shared goals. Articulating the benefits of the APCO Bone Health QI activity can help promote engagement and cooperation.
“The critical factors to success include having a supportive organisational culture, good design and planning, and follow through on results, ensuring the data collection is meaningful and achievable,” Dr Hew said.
“Data collected through the audit should be used to benchmark practice against the standards recommended by The APCO Framework. This will show stakeholders that the audit was worth the effort, and that it contributed to tangible change.”
“It is hoped that lessons from implementing these clinical standards at the institutional level will be key to achieving change at the national level, through the development, or revision of national guidelines, and policy change,” said Dr Philippe Halbout, CEO, International Osteoporosis Foundation, Switzerland.
“These coordinated systems of care aim to identify, treat and monitor patients with osteoporosis. Timely intervention can reduce fractures by up to 50 per cent, deliver substantial financial savings, and ultimately, save lives.”
Osteoporosis is greatly under-diagnosed and under-treated in Asia, even among those at highest risk who have already fractured.6
The APCO Bone Health QI Tool Kit focuses on closing this gap by:
* Understanding the baseline variables and current state of osteoporosis care in any given institution or health care practice.
* Enabling benchmarking clinical practice against The APCO Framework, evidence-based clinical practice guidelines or established protocols for the chosen standard of care.
* Using the findings to then improve the quality of care through iterative PDSA cycles.
* Using the lessons learned from implementation of the APCO Clinical Standard at an institutional level, to build momentum and work towards inclusion of the quality improvement/clinical standard in national guidelines, and lobbying for funding and policy change, as appropriate.
Practically, PDSA cycles are a quality improvement strategy that tests a change on a small scale. The PDSA builds on the learning from test cycles in a structured way before implementation on a wider scale.7
The PDSA cycles are iterative. Each cycle involves the following four stages:
1. Plan — developing a plan to test the change;
2. Do — carrying out the test;
3. Study — observing and learning; and
4. Act — planning the next change cycle or full implementation.
“Assessment of adherence to pharmacological treatments that are recommended in new or revised osteoporosis clinical guidelines should be undertaken on an ongoing basis, after initiation of therapy, and appropriate corrective action can be taken if treated individuals have become non-adherent,” Dr Chandran advised.
Every patient living with osteoporosis and/or a fragility fracture passes through several points on their journey from diagnosis, through to treatment and follow-up. The interface of this pathway with other entities, for example, referrals to other care givers or departments, post- discharge follow-ups, are also important considerations in the quality of care received.
“The launch of the APCO Bone Health QI Tool Kit demonstrates the organisation’s ongoing commitment to achieving better care for patients with greater consistency in national and regional clinical practice guidelines for the screening, diagnosis, and management of osteoporosis in the Asia Pacific,” said Dr Halbout.
To learn more about the APCO Bone Health QI Tool Kit, visit www.apcobonehealth.org or follow APCO on LinkedIn: asia-pacific-consortium-on-osteoporosis.
About the APCO Bone Health QI Tool Kit
APCO developed the Bone Health QI Tool Kit to enable healthcare providers worldwide, to benchmark their practices against the clinical standards proposed in The APCO Framework. The tool kit provides guidance on undertaking and implementing quality improvement (QI) projects centred around seven key clinical standards for the screening, diagnosis, and management of osteoporosis.
With the key objective of promoting QI of osteoporosis care, the APCO audit focuses on investigating the patient pathway, identifying the gaps, or missed opportunities for patients at risk, and formulating recommendations to address these gaps. Implementation of the QI will be through Plan-Do-Study-Act (PDSA) cycles – a quality improvement strategy that tests a change on a small scale, before implementation on a wider scale. The tool kit offers practical tips on conducting an audit, that generates meaningful data, that can lead ultimately to implementing changes in clinical practices.
About APCO
The Asia Pacific Consortium on Osteoporosis (APCO) comprises 46 osteoporosis experts from 20 countries and regions, charged with developing tangible solutions to the substantive challenges involving osteoporosis management and fracture prevention in this most populated and fastest growing part of the world. APCO’s mission is to engage with relevant stakeholders, including healthcare providers, policy makers and the public, to help develop and implement country and region-specific programs for the prevention and treatment of osteoporosis, and its complication of fragility fractures, in the Asia Pacific. http://www.apcobonehealth.org
About osteoporosis in the Asia Pacific region
* Osteoporosis – the most common disease – is characterised by porous and brittle bones whose density and quality are poor, resulting in structural skeletal deterioration.1
* The Asia Pacific is home to 4.5 billion people with vastly different healthcare systems.8
* In most developing countries of the Asia Pacific, especially in rural areas, DXA technology is not widely available.9
* Low levels of vitamin D are observed region-wide in the Asia Pacific. This can be attributed to several causal factors, including genetic, lifestyle, seasonal and geographical influences.10
* More than 68 per cent of the countries in the IOF’s Asia Pacific’s Regional Audit now have populations that are over 50 per cent urban, and the resulting reduction in sunlight exposure due to pollution and overcrowding, is affecting vitamin D levels, which contribute to osteoporosis risk.10
* Nearly all Asian countries fall far below The Food and Agriculture Organization of the United Nations / World Health Organization (WHO) recommendations for calcium intake of between 1,000 and 1,300 mg/day. The calcium intake for Malaysia, the Philippines and India remains below 500 mg/day.10
DIGITAL MEDIA KIT : http://www.apcotoolkit.org
Asia Pacific Consortium on Osteoporosis (APCO)
secretariat@apcobonehealth.org
References
1. International Osteoporosis Foundation (IOF). Capture the fracture. [Oct, 2021]; Available from: https://www.capturethefracture.org/about
2. Chandran M, et al., Development of the Asia Pacific Consortium on Osteoporosis (APCO) Framework: clinical standards of care for the screening, diagnosis, and management of osteoporosis in the Asia-Pacific region. Osteoporos Int., 2021. 32(7): p. 1249-1275.
3. Cheung, C.-L., et al., An updated hip fracture projection in Asia: The Asian Federation of Osteoporosis Societies study. Osteoporosis and sarcopenia, 2018. 4(1): p. 16-21.
4. Chandran, M., Ebeling, P., Mitchell, P., Nguyen, T., Harmonization of Osteoporosis Guidelines: Paving the Way for Disrupting the Status Quo in Osteoporosis Management in the Asia Pacific. JBMR, 2022.
5. Watts, J., Ambimanyi-Ochom, J, & Sander K, Osteoporosis costing all Australians: A new burden of disease analysis 2012-2022. 2013, Osteoporosis Australia.
6. International Osteoporosis Foundation (IOF). Key statistics for Asia. [cited October 2021]; https://www.osteoporosis.foundation/facts-statistics/key-statistic-for-asia
7. NHS England and NHS Improvement. Plan, Do, Study, Act (PDSA) cycles and the model for improvement. https://www.england.nhs.uk/wp-content/uploads/2022/01/qsir-pdsa-cycles-model-for-improvement.pdf
8. Wang, P., et al., Estimation of Prevalence of Osteoporosis Using OSTA and Its Correlation with Sociodemographic Factors, Disability and Comorbidities. International journal of environmental research and public health, 2019. 16(13): p. 2338.
9. Chandran, M., et al., IQ driving QI: the Asia Pacific Consortium on Osteoporosis (APCO): an innovative and collaborative initiative to improve osteoporosis care in the Asia Pacific. Osteoporos Int, 2020. 31(11): p. 2077-2081.
10. International Osteoporosis Foundation (IOF). The Asia-Pacific Regional Audit. 2013 [cited May 2022]; https://www.osteoporosis.foundation/sites/iofbonehealth/files/2019-06/2013_Asia_Pacific_Audit_English.pdf
Copyright 2022 ACN Newswire. All rights reserved. http://www.acnnewswire.com
The APCO Bone Health QI Tool Kit – Putting a brake on fractures in the world’s most populous & fastest ageing region
8TAIPEI, May 24 – (ACN Newswire) – An osteoporotic fracture occurs every three seconds worldwide.1 With more than 50 per cent of the world’s hip fractures expected to occur in the Asia Pacific (AP) region over the next two decades, the Asia Pacific Consortium on Osteoporosis (APCO) has launched a Bone Health QI Tool Kit today (May 24, 2022), to promote quality improvement (QI) of osteoporosis care.
This comprehensive tool kit, that encompasses multiple components in the screening, diagnosis and management of osteoporosis, is a world-first of its kind, said Dr Manju Chandran, APCO Chairperson and Director of the Osteoporosis and Bone Metabolism Unit, Singapore General Hospital, Singapore.
“Previous audits have focused on just a single aspect of osteoporosis care, namely the identification and appropriate management of osteoporotic fragility fractures. A perfect storm of a rise in osteoporosis among Asia-Pacific populations is expected to occur due to the region’s rapidly ageing population, mounting urbanisation, and subsequent increase in sedentary lifestyles,”2 Dr Chandran said.
“A recent analysis of nine Asian countries and regions reveals the number of hip fractures will more than double from 1.13 million in 2018, to 2.54 million in 2050,3 primarily due to changing population demographics.”
The APCO Bone Health QI Tool Kit is based on seven selected standards from The APCO Framework that apply to the clinical setting. Implementation of the QI will be through iterative Plan-Do-Study-Act (PDSA) cycles.
“Once the audits are completed, healthcare professionals (HCPs) and medical centres can commence implementing changes to their clinical practice. APCO is in the process of engaging QI experts to help train APCO members to perform such projects in their individual medical centres and practices,” said Dr Chandran.
The tool kit will enable these auditing exercises to be undertaken to establish levels of adherence with those standards of care within The APCO Framework that can be implemented directly into clinical practice.4
“Given the current increasing incidence of osteoporosis amongst our population, our country now has the highest prevalence of hip fracture of any country in Asia, which is also higher than the world’s average,”6 said Professor Derrick Chan, APCO Committee member and Director, Department of Geriatrics and Gerontology, National Taiwan University Hospital (NTUH), Chinese Taipei.
“The likelihood of successfully creating change to stem this bone health crisis will rest largely upon the efforts of HCPs to engage their peers to work towards shared goals. Articulating the benefits of the APCO Bone Health QI activity can help promote engagement and cooperation.
“The critical factors to success include having a supportive organisational culture, good design and planning, and follow through on results, ensuring the data collection is meaningful and achievable,” Professor Chan said.
“Data collected through the audit should be used to benchmark practice against the standards recommended by The APCO Framework. This will show stakeholders that the audit was worth the effort, and that it contributed to tangible change.”
The APCO audit focuses on investigating the patient pathway, identifying the gaps or missed opportunities for patients at risk, and then the PDSA activity is used to improve quality of care and to formulate recommendations.
“It is hoped that lessons from implementing these clinical standards at the institutional level will be key to achieving change at the national level, through the development, or revision of national guidelines, and policy change,” said Dr Philippe Halbout, CEO, International Osteoporosis Foundation, Switzerland.
“These coordinated systems of care aim to identify, treat and monitor patients with osteoporosis. Timely intervention can reduce fractures by up to 50 per cent, deliver substantial financial savings, and ultimately, save lives.”
Osteoporosis is greatly under-diagnosed and under-treated in Asia, even among those at highest risk who have already fractured.6
The APCO Bone Health QI Tool Kit focuses on closing this gap by:
* Understanding the baseline variables and current state of osteoporosis care in any given institution or health care practice.
* Enabling benchmarking clinical practice against The APCO Framework, evidence-based clinical practice guidelines or established protocols for the chosen standard of care.
* Using the findings to then improve the quality of care through iterative PDSA cycles.
* Using the lessons learned from implementation of the APCO Clinical Standard at an institutional level, to build momentum and work towards inclusion of the quality improvement/clinical standard in national guidelines, and lobbying for funding and policy change, as appropriate.
Practically, PDSA cycles are a quality improvement strategy that tests a change on a small scale. The PDSA builds on the learning from test cycles in a structured way before implementation on a wider scale.7
The PDSA cycles are iterative. Each cycle involves the following four stages:
1. Plan — developing a plan to test the change;
2. Do — carrying out the test;
3. Study — observing and learning; and
4. Act — planning the next change cycle or full implementation.
“Assessment of adherence to pharmacological treatments that are recommended in new or revised osteoporosis clinical guidelines should be undertaken on an ongoing basis, after initiation of therapy, and appropriate corrective action can be taken if treated individuals have become non-adherent,” Dr Chandran advised.
Every patient living with osteoporosis and/or a fragility fracture passes through several points on their journey from diagnosis, through to treatment and follow-up. The interface of this pathway with other entities, for example, referrals to other care givers or departments, post- discharge follow-ups, are also important considerations in the quality of care received.
“The launch of the APCO Bone Health QI Tool Kit demonstrates the organisation’s ongoing commitment to achieving better care for patients with greater consistency in national and regional clinical practice guidelines for the screening, diagnosis, and management of osteoporosis in the Asia Pacific,” said Dr Halbout.
To learn more about the APCO Bone Health QI Tool Kit, visit www.apcobonehealth.org or follow APCO on LinkedIn: asia-pacific-consortium-on-osteoporosis.
About the APCO Bone Health QI Tool Kit
APCO developed the Bone Health QI Tool Kit to enable healthcare providers worldwide, to benchmark their practices against the clinical standards proposed in The APCO Framework. The tool kit provides guidance on undertaking and implementing quality improvement (QI) projects centred around seven key clinical standards for the screening, diagnosis, and management of osteoporosis.
With the key objective of promoting QI of osteoporosis care, the APCO audit focuses on investigating the patient pathway, identifying the gaps, or missed opportunities for patients at risk, and formulating recommendations to address these gaps. Implementation of the QI will be through Plan-Do-Study-Act (PDSA) cycles – a quality improvement strategy that tests a change on a small scale, before implementation on a wider scale. The tool kit offers practical tips on conducting an audit, that generates meaningful data, that can lead ultimately to implementing changes in clinical practices.
About APCO
The Asia Pacific Consortium on Osteoporosis (APCO) comprises 46 osteoporosis experts from 20 countries and regions, charged with developing tangible solutions to the substantive challenges involving osteoporosis management and fracture prevention in this most populated and fastest growing part of the world. APCO’s mission is to engage with relevant stakeholders, including healthcare providers, policy makers and the public, to help develop and implement country and region-specific programs for the prevention and treatment of osteoporosis, and its complication of fragility fractures, in the Asia Pacific. http://www.apcobonehealth.org
About osteoporosis in the Asia Pacific region
* Osteoporosis – the most common disease – is characterised by porous and brittle bones whose density and quality are poor, resulting in structural skeletal deterioration.1
* The Asia Pacific is home to 4.5 billion people with vastly different healthcare systems.8
* In most developing countries of the Asia Pacific, especially in rural areas, DXA technology is not widely available.9
* Low levels of vitamin D are observed region-wide in the Asia Pacific. This can be attributed to several causal factors, including genetic, lifestyle, seasonal and geographical influences.10
* More than 68 per cent of the countries in the IOF’s Asia Pacific’s Regional Audit now have populations that are over 50 per cent urban, and the resulting reduction in sunlight exposure due to pollution and overcrowding, is affecting vitamin D levels, which contribute to osteoporosis risk.10
* Nearly all Asian countries fall far below The Food and Agriculture Organization of the United Nations / World Health Organization (WHO) recommendations for calcium intake of between 1,000 and 1,300 mg/day. The calcium intake for Malaysia, the Philippines and India remains below 500 mg/day.10
DIGITAL MEDIA KIT : http://www.apcotoolkit.org
Asia Pacific Consortium on Osteoporosis (APCO)
secretariat@apcobonehealth.org
References
1. International Osteoporosis Foundation (IOF). Capture the fracture. [Oct, 2021]; Available from: https://www.capturethefracture.org/about.
2. Chandran M, et al., Development of the Asia Pacific Consortium on Osteoporosis (APCO) Framework: clinical standards of care for the screening, diagnosis, and management of osteoporosis in the Asia-Pacific region. Osteoporos Int., 2021. 32(7): p. 1249-1275.
3. Cheung, C.-L., et al., An updated hip fracture projection in Asia: The Asian Federation of Osteoporosis Societies study. Osteoporosis and sarcopenia, 2018. 4(1): p. 16-21.
4. Chandran, M., Ebeling, P., Mitchell, P., Nguyen, T., Harmonization of Osteoporosis Guidelines: Paving the Way for Disrupting the Status Quo in Osteoporosis Management in the Asia Pacific. JBMR, 2022.
5. Watts, J., Ambimanyi-Ochom, J, & Sander K, Osteoporosis costing all Australians: A new burden of disease analysis 2012-2022. 2013, Osteoporosis Australia.
6. International Osteoporosis Foundation (IOF). Key statistics for Asia. [cited October 2021]; Available from: https://www.osteoporosis.foundation/facts-statistics/key-statistic-for-asia.
7. NHS England and NHS Improvement. Plan, Do, Study, Act (PDSA) cycles and the model for improvement. From: https://www.england.nhs.uk/wp-content/uploads/2022/01/qsir-pdsa-cycles-model-for-improvement.pdf.
8. Wang, P., et al., Estimation of Prevalence of Osteoporosis Using OSTA and Its Correlation with Sociodemographic Factors, Disability and Comorbidities. International journal of environmental research and public health, 2019. 16(13): p. 2338.
9. Chandran, M., et al., IQ driving QI: the Asia Pacific Consortium on Osteoporosis (APCO): an innovative and collaborative initiative to improve osteoporosis care in the Asia Pacific. Osteoporos Int, 2020. 31(11): p. 2077-2081.
10. International Osteoporosis Foundation (IOF). The Asia-Pacific Regional Audit. 2013 [cited May 2022]; Available from: https://www.osteoporosis.foundation/sites/iofbonehealth/files/2019-06/2013_Asia_Pacific_Audit_English.pdf.
Copyright 2022 ACN Newswire. All rights reserved. http://www.acnnewswire.com
The APCO Bone Health QI Tool Kit – Putting a brake on fractures in the world’s most populous & fastest ageing region

SINGAPORE, May 24, 2022 – (ACN Newswire) – An osteoporotic fracture occurs every three seconds worldwide.1 With more than 50 per cent of the world's hip fractures expected to occur in the Asia Pacific (AP) region over the next two decades, the Asia Pacific Consortium on Osteoporosis (APCO) has launched a Bone Health QI Tool Kit today (May 24, 2022), to promote quality improvement (QI) of osteoporosis care.
 |
This comprehensive tool kit, that encompasses multiple components in the screening, diagnosis and management of osteoporosis, is a world-first of its kind, said Dr Manju Chandran, APCO Chairperson and Director of the Osteoporosis and Bone Metabolism Unit, Singapore General Hospital, Singapore.
"Previous audits have focused on just a single aspect of osteoporosis care, namely the identification and appropriate management of osteoporotic fragility fractures. A perfect storm of a rise in osteoporosis among Asia-Pacific populations is expected to occur due to the region's rapidly ageing population, mounting urbanisation, and subsequent increase in sedentary lifestyles,"2 Dr Chandran said.
"A recent analysis of nine Asian countries and regions reveals the number of hip fractures will more than double from 1.13 million in 2018, to 2.54 million in 2050,3 primarily due to changing population demographics."
The APCO Bone Health QI Tool Kit is based on seven selected standards from The APCO Framework that apply to the clinical setting. Implementation of the QI will be through iterative Plan-Do-Study-Act (PDSA) cycles.
"Once the audits are completed, healthcare professionals (HCPs) and medical centres can commence implementing changes to their clinical practice. APCO is in the process of engaging QI experts to help train APCO members to perform such projects in their individual medical centres and practices," said Dr Chandran.
The tool kit will enable these auditing exercises to be undertaken to establish levels of adherence with those standards of care within The APCO Framework that can be implemented directly into clinical practice.4
This year in Australia, an estimated 6.2 million people aged over 50 years will be living with osteoporosis or osteopenia, representing a 31 per cent increase from 2012,5 said Dr Greg Lyubomirsky, CEO, Healthy Bones Australia.
"The likelihood of successfully creating change to stem this bone health crisis will rest largely upon the efforts of HCPs to engage their peers to work towards shared goals. Articulating the benefits of the APCO Bone Health QI activity can help promote engagement and cooperation.
"The critical factors to success include having a supportive organisational culture, good design and planning, and follow through on results, ensuring the data collection is meaningful and achievable," Dr Lyubomirsky said.
"Data collected through the audit should be used to benchmark practice against the standards recommended by The APCO Framework. This will show stakeholders that the audit was worth the effort, and that it contributed to tangible change."
The APCO audit focuses on investigating the patient pathway, identifying the gaps or missed opportunities for patients at risk, and then the PDSA activity is used to improve quality of care and to formulate recommendations.
"It is hoped that lessons from implementing these clinical standards at the institutional level will be key to achieving change at the national level, through the development, or revision of national guidelines, and policy change," said Dr Philippe Halbout, CEO, International Osteoporosis Foundation, Switzerland.
"These coordinated systems of care aim to identify, treat and monitor patients with osteoporosis. Timely intervention can reduce fractures by up to 50 per cent, deliver substantial financial savings, and ultimately, save lives."
Osteoporosis is greatly under-diagnosed and under-treated in Asia, even among those at highest risk who have already fractured.6
The APCO Bone Health QI Tool Kit focuses on closing this gap by:
— Understanding the baseline variables and current state of osteoporosis care in any given institution or health care practice.
— Enabling benchmarking clinical practice against The APCO Framework, evidence-based clinical practice guidelines or established protocols for the chosen standard of care.
— Using the findings to then improve the quality of care through iterative PDSA cycles.
— Using the lessons learned from implementation of the APCO Clinical Standard at an institutional level, to build momentum and work towards inclusion of the quality improvement/clinical standard in national guidelines, and lobbying for funding and policy change, as appropriate.
Practically, PDSA cycles are a quality improvement strategy that tests a change on a small scale. The PDSA builds on the learning from test cycles in a structured way before implementation on a wider scale.7
The PDSA cycles are iterative. Each cycle involves the following four stages:
1. Plan — developing a plan to test the change;
2. Do — carrying out the test;
3. Study — observing and learning; and
4. Act — planning the next change cycle or full implementation.
"Assessment of adherence to pharmacological treatments that are recommended in new or revised osteoporosis clinical guidelines should be undertaken on an ongoing basis, after initiation of therapy, and appropriate corrective action can be taken if treated individuals have become non-adherent," Dr Chandran advised.
Every patient living with osteoporosis and/or a fragility fracture passes through several points on their journey from diagnosis, through to treatment and follow-up. The interface of this pathway with other entities, for example, referrals to other care givers or departments, post- discharge follow-ups, are also important considerations in the quality of care received.
"The launch of the APCO Bone Health QI Tool Kit demonstrates the organisation's ongoing commitment to achieving better care for patients with greater consistency in national and regional clinical practice guidelines for the screening, diagnosis, and management of osteoporosis in the Asia Pacific," said Dr Halbout.
To learn more about the APCO Bone Health QI Tool Kit,
visit www.apcobonehealth.org or follow APCO on LinkedIn: asia-pacific-consortium-on-osteoporosis.
About the APCO Bone Health QI Tool Kit
APCO developed the Bone Health QI Tool Kit to enable healthcare providers worldwide, to benchmark their practices against the clinical standards proposed in The APCO Framework. The tool kit provides guidance on undertaking and implementing quality improvement (QI) projects centred around seven key clinical standards for the screening, diagnosis, and management of osteoporosis.
With the key objective of promoting QI of osteoporosis care, the APCO audit focuses on investigating the patient pathway, identifying the gaps, or missed opportunities for patients at risk, and formulating recommendations to address these gaps. Implementation of the QI will be through Plan-Do-Study-Act (PDSA) cycles – a quality improvement strategy that tests a change on a small scale, before implementation on a wider scale. The tool kit offers practical tips on conducting an audit, that generates meaningful data, that can lead ultimately to implementing changes in clinical practices.
About APCO
The Asia Pacific Consortium on Osteoporosis (APCO) comprises 46 osteoporosis experts from 20 countries and regions, charged with developing tangible solutions to the substantive challenges involving osteoporosis management and fracture prevention in this most populated and fastest growing part of the world. APCO's mission is to engage with relevant stakeholders, including healthcare providers, policy makers and the public, to help develop and implement country and region-specific programs for the prevention and treatment of osteoporosis, and its complication of fragility fractures, in the Asia Pacific. www.apcobonehealth.org
About osteoporosis in the Asia Pacific region
* Osteoporosis – the most common disease – is characterised by porous and brittle bones whose density and quality are poor, resulting in structural skeletal deterioration.1
* The Asia Pacific is home to 4.5 billion people with vastly different healthcare systems.8
* In most developing countries of the Asia Pacific, especially in rural areas, DXA technology is not widely available.9
* Low levels of vitamin D are observed region-wide in the Asia Pacific. This can be attributed to several causal factors, including genetic, lifestyle, seasonal and geographical influences.10
* More than 68 per cent of the countries in the IOF's Asia Pacific's Regional Audit now have populations that are over 50 per cent urban, and the resulting reduction in sunlight exposure due to pollution and overcrowding, is affecting vitamin D levels, which contribute to osteoporosis risk.10
* Nearly all Asian countries fall far below The Food and Agriculture Organization of the United Nations / World Health Organization (WHO) recommendations for calcium intake of between 1,000 and 1,300 mg/day. The calcium intake for Malaysia, the Philippines and India remains below 500 mg/day.10
DIGITAL MEDIA KIT: www.apcotoolkit.org
Asia Pacific Consortium on Osteoporosis (APCO)
secretariat@apcobonehealth.org
References
1. International Osteoporosis Foundation (IOF). Capture the fracture. [Oct, 2021]; Available from: https://www.capturethefracture.org/about.
2. Chandran M, et al., Development of the Asia Pacific Consortium on Osteoporosis (APCO) Framework: clinical standards of care for the screening, diagnosis, and management of osteoporosis in the Asia-Pacific region. Osteoporos Int., 2021. 32(7): p. 1249-1275.
3. Cheung, C.-L., et al., An updated hip fracture projection in Asia: The Asian Federation of Osteoporosis Societies study. Osteoporosis and sarcopenia, 2018. 4(1): p. 16-21.
4. Chandran, M., Ebeling, P., Mitchell, P., Nguyen, T., Harmonization of Osteoporosis Guidelines: Paving the Way for Disrupting the Status Quo in Osteoporosis Management in the Asia Pacific. JBMR, 2022.
5. Watts, J., Ambimanyi-Ochom, J, & Sander K, Osteoporosis costing all Australians: A new burden of disease analysis 2012-2022. 2013, Osteoporosis Australia.
6. International Osteoporosis Foundation (IOF). Key statistics for Asia. [cited October 2021]; Available from: https://www.osteoporosis.foundation/facts-statistics/key-statistic-for-asia.
7. NHS England and NHS Improvement. Plan, Do, Study, Act (PDSA) cycles and the model for improvement. From: https://www.england.nhs.uk/wp-content/uploads/2022/01/ qsir-pdsa-cycles-model-for-improvement.pdf.
8. Wang, P., et al., Estimation of Prevalence of Osteoporosis Using OSTA and Its Correlation with Sociodemographic Factors, Disability and Comorbidities. International Journal of Environmental Research and Public Health, 2019. 16(13): p. 2338.
9. Chandran, M., et al., IQ driving QI: the Asia Pacific Consortium on Osteoporosis (APCO): an innovative and collaborative initiative to improve osteoporosis care in the Asia Pacific. Osteoporos Int, 2020. 31(11): p. 2077-2081.
10. International Osteoporosis Foundation (IOF). The Asia-Pacific Regional Audit. 2013 [cited May 2022]; From: https://www.osteoporosis.foundation/sites/iofbonehealth/files/2019-06/ 2013_Asia_Pacific_Audit_English.pdf
Copyright 2022 ACN Newswire. All rights reserved. http://www.acnnewswire.com
Malaysian Genomics Resource Centre Berhad Posts 212% Increase in Revenue for 3Q FY2022

PETALING JAYA, Malaysia, May 23, 2022 – (ACN Newswire) – Malaysian Genomics Resource Centre Berhad, a leading genomics and biopharmaceutical specialist, today announced that the Group registered a 211.66% increase in revenue to RM5.08 million for the third quarter ended 31 March 2022 (3Q 2022) compared with revenue of RM1.63 million for the same quarter of the previous financial year.
 |
For the quarter under review, the Group posted profit before tax (PBT) of RM1.96 million compared with PBT of RM0.2 million in 3Q 2021. Earnings per share (EPS) stood at 1.60 sen for 3Q 2022 compared with EPS of 0.19 sen in the corresponding quarter of the previous financial year.
For the nine months ended 31 March 2022 (9M FY2022), Malaysian Genomics registered a nearly 12-fold increase in revenue to RM22.03 million compared with RM1.85 million in 9M FY2021. The Group registered RM3.21 million in PBT for 9M FY2022 compared with a loss before tax of RM2.2 million in the corresponding period of the preceding financial year.
Encik Noor Azri bin Dato' Sri Noor Azerai (Azri Azerai), Executive Director of Malaysian Genomics, said, "The improvement in revenue for the quarter under review was due to the Group's diversification into biopharmaceutical services, primarily the distribution of immunotherapy and cell therapies, together with higher demand for genetic screening services following the reopening of the economy. COVID-19 vaccine administration and distribution contributed to our revenue growth as well."
"Our PBT was supported by higher margin from the Group's successful and ongoing penetration into the distribution of immunotherapy and cell therapies. We continue to seek partnerships and opportunities in which we can have a wider market reach for our services, while our laboratory's Current Good Manufacturing Practice approval will add value to our capabilities, including the production of CAR T-cells for solid cancers as well as the contract manufacturing of other types of cells for third-parties that will generate new revenue streams."
Malaysian Genomics Resource Centre Berhad: http://www.mgrc.com.my/
Malaysian Genomics Resource Centre Berhad: 0155 / [BURSA: MGRC] [RIC: MGRC:KL] [BBG: MGRC:MK]
Copyright 2022 ACN Newswire. All rights reserved. http://www.acnnewswire.com
